Nature of electricity is amazing. Electricity is among the top 10 needs of everybody as people are more onto electric current powered lifestyles these days. I’ve talked about electric current in a post or two earlier, and this post too is going to be about electricity. It’s about effects of electric current.
There are 3 main effects of electricity as,
- Heating effect
- Magnetic effect (Magnetism)
- Chemical effect
Let me explain how these 3 electric effects work and their benefits to us in the next sections.
Heating Effect of Electric Current
As this electric effect’s name imply, generation of heat due to electric current is known as the heating effect of electricity. Don’t want to believe it? Well, it’s time for a simple experiment.
Take 2 cm length of thin Nichrome wire coil and connect it to a battery and a key as in the image. Press the key for about 30 seconds. Now touch the Nichrome wire coil. What will you feel? Would it be warm or cool? Obviously it’ll be warm.
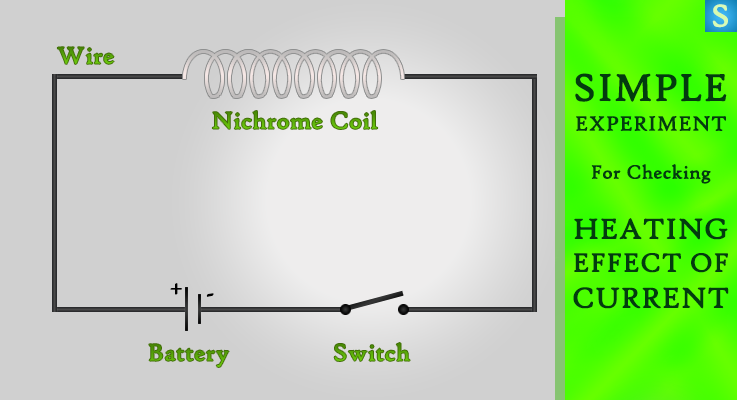
It’s clear that the wire is heated because the flow of electric current through the wire. When electrons pass through a wire, they can give some of their energy to the atoms in the wire and make them vibrate more faster than their original vibration, so that the wire is heated. We can define that phenomenon as the heating effect of electric current.
You can increase the heating effect of current. First let me tell how to reveal what factors we have to change for that.
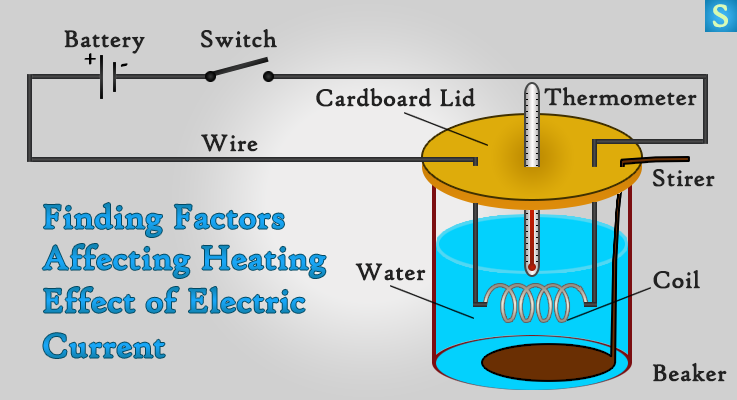
Experiment to find factors affecting Heating Effect
1. Electric Current
First factor is how electric current increase can also improve the heating effect of current.
Take a small beaker half filled with water and cover it with a cardboard lid. Insert a 2 cm length Nichrome wire coil into the water as well as a stirrer. Then connect the ends of the Nichrome coil to a circuit with a battery and a switch.
Place a thermometer into the water, so that you can check the water temperature. Close the circuit with the help of the switch and stir the water. Keep it that way for like 5 minutes and then measure the temperature of water.
Repeat the same thing by using two batteries and three batteries.
When you add more batteries to above circuit, the electric current increases. Temperature readings will be low, high and very high in each stage in the above experiment respectively. It’s clear that as the electric current increases, the heating effect also increases, while a decrease in electric current also decreases the heating effect.
2. Resistance
The next factor is the resistance of the Nichrome wire coil. Let’s use the same apparatus with a slight modification.
Replace the 2 cm Nichrome coil with 4 cm Nichrome coil. Close the circuit. Stir the water. Measure the temperature of water at each battery stage. Now compare these temperature readings with the previous experiment’s temperature readings.
You’ll notice that the temperature of water is greater than that in the first experiment for all 3 stages. As you can figure it out, once the resistance of the Nichrome coil increases (as its length increases), it is clear that the heating effect too increases as the resistance increases.
Conductor Resistance by Length
Wires are electrical conductors same as metals. Let’s see how variation of resistance occur as a conductor’s length increases.
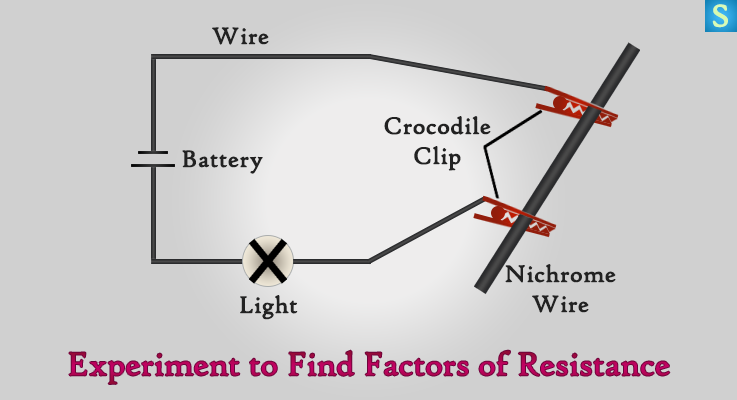
Connect the circuit as shown with 2 crocodile clips at A and B points with a battery and light bulb. You need 30 cm of gauge 30 Nichrome wire to complete the circuit.
- Keep the crocodile clips closer on the wire and then notice the brightness of the light bulb.
- Now move the clips further apart and compare the brightness of the light bulb with the above.
You’ll realize that the brightness of the light bulb increases, when the crocodile clips are closer and the brightness decreases as the clips move apart.
The brightness of the bulb decreases due to the resistance of the wire increases as the length increases, while the brightness of the bulb increases when the resistance of the wire decreases as the length decreases.
Conductor Resistance by Cross-section Area
Using the same circuit explained above, keep the crocodile clips on the wire 20 cm apart and notice the brightness of the light bulb. Now add another identical Nichrome wire between the crocodile clips so that the area of cross section is doubled.
Then compare the brightness of the light bulb with the previous brightness.
You can see here that the brightness of the bulb increases when using two Nichrome wires instead of one wire. The area of cross section of the conductor increases, when you use two wires. Hence the brightness of the bulb increases thanks to reduction of resistance.
Conductor Resistance by Material
It’s time to find how the resistance of the conductor depends on its material.
You can use the same circuit used in the last 2 experiments for this one. Apart the crocodile clips by 20 cm on the wire and observe the brightness of the light bulb. Switch the Nichrome wire with a Copper wire of the same length and same cross sectional area (gauge 30 in this case) and again observe the brightness of the bulb.
Brightness of the bulb is not the same in each case. Therefore you can come to the conclusion that the brightness of the light bulb changes, due to the resistance of the conductor differs based on the conductor material despite the length and area of cross section are the same.
3. Time of Passing Current
Yeah, the third factor is time. Repeat the either of experiment #1 or #2 by using the same Nichrome coil, Voltage supply while specifically varying the time of passing current. For example, previously it was after 5 mins, so measure the water temperature after 10-15 mins this time.
You can observe here that the water temperature increases as the time of passing current increases. Therefore it is clear that the heating effect of electric current too increases as the time of passing current increases.
Electric Energy Formula Looks Like …
You can calculate the electric energy using the below formula. It’s created based on the factors that affect the heating effect of electric current as discovered through a series of experiments explained above.
| Supplied Voltage | = V (Volts) |
| Current through the coil | = I (Ampere) |
| Resistance of the coil | = R (Ohms) |
| Time of passing current | = t (Seconds) |
| Electrical energy transmitted | = VIt = I2Rt = V2t/R (Joules) |
| E = VIt (Joules) | |
Heating Effect Benefits
As you may have guessed, heating stuffs is the benefit of heating effect of electric current. Simply you’re converting electrical energy to heat energy thanks to this effect of current.
- Fuse is a basic electrical part used to protect appliances. Fuse wire melts due to the heating effect of current when the current flowing through is exceeded than the rated amperage.
- Water heaters both in Kitchen & Bathroom uses this effect of current.
- Light bulbs also works due to the heating effect. Once the filament is heated by electricity, it starts glowing.
- All kinds of Irons such as Clothes Iron, Hair Irons, Soldering Irons all work by using the heating effect of current.
Here are some home appliances which use the heating effect of the current. Which of these do you have in your home? Have you used them before learning about this effect of current? Comment below.
- Electric Kettle
- Electric Cooker
- Electric Oven
- Toaster
- Water Heater
- Clothes Iron
- Light Bulbs

It’s easy to get yourself hurt while using electricity, unless you know these handy electrical safety precautions.
Electricity and Magnetism (Magnetic Effect)
The next effect of electric current is Magnetism. In 1819, Hans Christian Oersted discovered the electricity has a magnetic effect. This experiment will help you understand the magnetic effect of electric current.
What you need,
- Compass
- Battery & Switch
- Wire
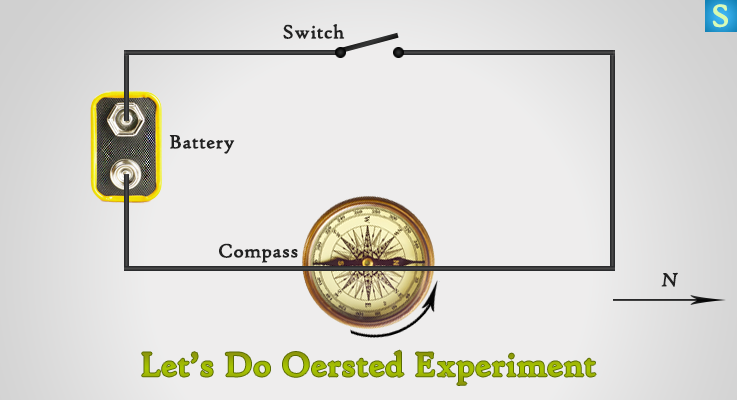
Create a circuit with a battery and a switch. You also need a compass for this experiment. Hold the wire on top of the compass needle. Now close the circuit with the switch for a couple of seconds. When current flows through the circuit, notice what happens to the compass needle.
Okay, let’s reverse the direction of electric current and then notice the what happens to the compass needle again.
In the first case, you will see the compass needle deflect towards a specific direction. Then in the second case of reverse direction of current also deflect the compass needle, but to the opposite direction to the first case.
Deflection of the compass needle changes due to the formation of a magnetic field. Therefore it is clear that a magnetism occurs around a conductor when an electric current flows through it.
Magnetic Field Around a Bar Magnet
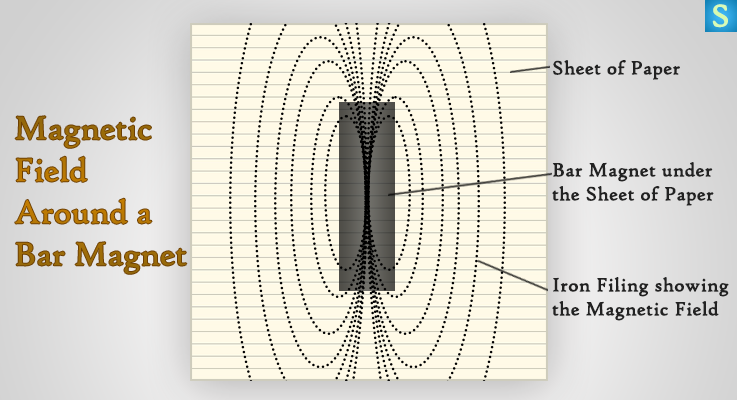
Place a thin glass or a sheet of paper on top of a bar magnet.
Sprinkle a thin layer of iron filings onto the paper or glass sheet. Slowly and gently tap the paper or glass sheet. Notice what is happening to the iron filings.
There are number of force lines around the bar magnet due to its magnetic field. When you tap the glass sheet or the paper, the iron filings point themselves along the lines of flux by acting like tiny compasses. Therefore the arrangement of iron fillings as shown in the image indicate the lines of force.
Magnetic Field Near Electric Current Passing Coil
Okay, now you have seen what happens around a bar magnet. What happens around an electric current passing conductor or a coil in this case? In the first Oersted Experiment, you learned that there’s a magnetic effect of current.
So the same thing happens around a bar magnet also happens around an electric current passing coil.
When an electric current passes through a long coil or a solenoid, a magnetic field is formed around it as shown in the image. This electromagnetic field is equivalent to a magnetic field around a bar magnet.
Therefore, a current carrying long coil or a solenoid act as a bar magnet which are known as electromagnets. The polarities of both ends of the solenoid changes according to the direction of electric current passes.
Factors Affecting Magnetism Strength of Electromagnets
Like anything else in this world, strength of Electromagnetism depends on a number of factors. They are,
- Magnitude of the electric current
- Number of turns of the coil
- Changing the soft iron core
The following experiment will help you understand how each of these 3 factors affect the electromagnetic strength.
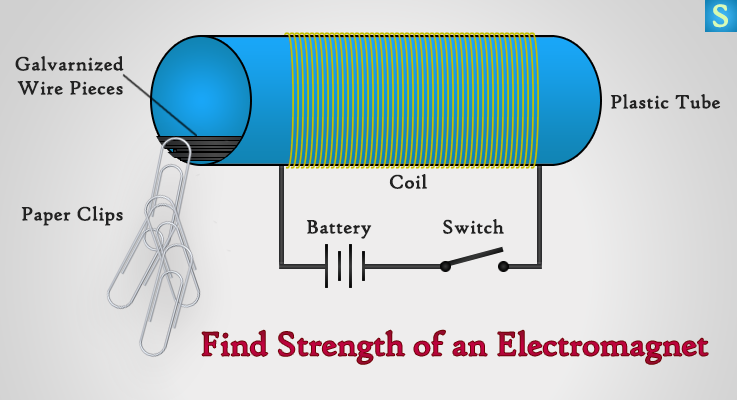
Take a small thread winding or any plastic tube, and wind 50 turns of insulated copper wire (30 swg) around the plastic tube. Here we’re using a Copper Wire because it flows electric current better and it doesn’t attract to magnets.
Cut a galvanized wire into 2 inch pieces and put a few pieces inside the plastic tube. Make sure to scrape the insulation of the wire pieces inside the tube using a sandpaper or a knife. Galvanized wire is usually made out of iron or steel which means, they get attracted to magnets.
Okay, let’s connect the copper coil to a battery and a switch and then close the circuit. Cool the electromagnet is live and let’s bring a couple of 1-2 paper clips chained together to one end of the galvanized wire pieces inside the tube. Bring more paper clips one by one and count the maximum number of paper clips that can be hung from the galvanized wire without falling.
- Repeat the above experiment by adding more batteries.
- Repeat again by increasing the number of turns up to 100.
- Repeat once more by inserting more pieces of galvanized wires.
You will see that the amount of paper clips that can be hung is always greater than the first instance as explained above.
The amount of clips which can be hung increases due to the fact that the field strength of the electromagnet increases. The magnitude of the electric current flowing through the coil increases as you add more batteries.
Therefore it is clear that the field strength of the electromagnet increases when,
- The current flowing through the coil increases
- The number of turns of the coil increases
- The amount of soft iron core increases
Uses of Electro Magnetic Effect of Current
Similar to the way people use heating effect of current, the magnetic effect of current is mostly used in home electrical appliances. That doesn’t mean it isn’t used in the apparatus of the industrial field. Okay?
Electromagnets are used in following situations,
- Electric Bells
- Circuit Breakers
- Electromagnetic Door Locks
- Electromagnetic Relays
- Telephone Ear Piece
- Cranes in Steel Works & Scrap Yards
Electromagnets are also used to remove splinters of steel or iron in hospitals dealing with eye injuries. Would you like to learn how exactly a Electric Bell works? Jump onto next section!
Electrical Bell – How It Works …
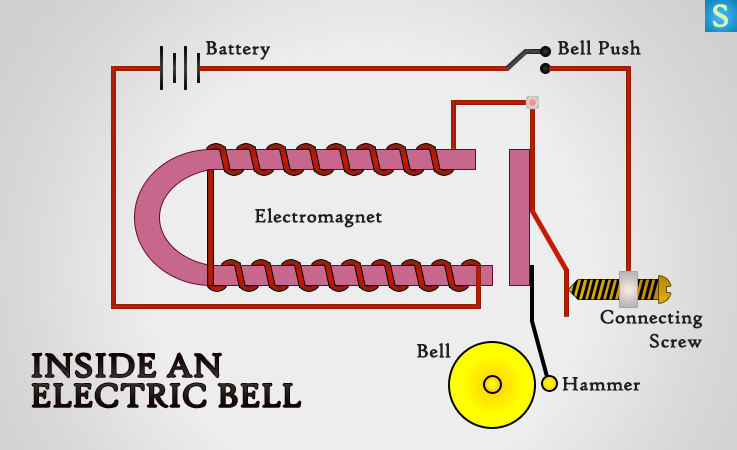
When you push bell switch, it completes the circuit of the bell and an electric current starts flowing through the coil. The U shaped soft-iron core becomes an electromagnet as a result of that and the soft-iron is attracted towards the electromagnet and the hammer hits the gong.
When the hammer attached soft-iron rod is attracted towards the electromagnet, circuit becomes no more closed, but opened.
So that the current stops flowing and switches off the electromagnet. The soft-iron rod goes back to its original state so that contact is made again, because of this process the bell will ring continuously as long as current is flowing.
Force on Current Passing Conductor in a Magnetic Field
What do you think would happen, if you were to pass an electric current through a conductor when the same environment is a magnetic field? Following experiment will reveal the answer.
You need,
- Horseshoe Magnet (powerful)
- Thin Copper Wire
- Battery & Switch
- Thread
Hang the thin copper wire between the poles of the horseshoe magnet using the thread. Connect the wire to the battery and the switch.
Stage #1: Switch ON and note the behavior of the wire.
Stage #2: Change the direction of electric current. Repeat it.
Stage #3: Turn the horseshoe magnet to the other side, so the polarity changes. Repeat it.
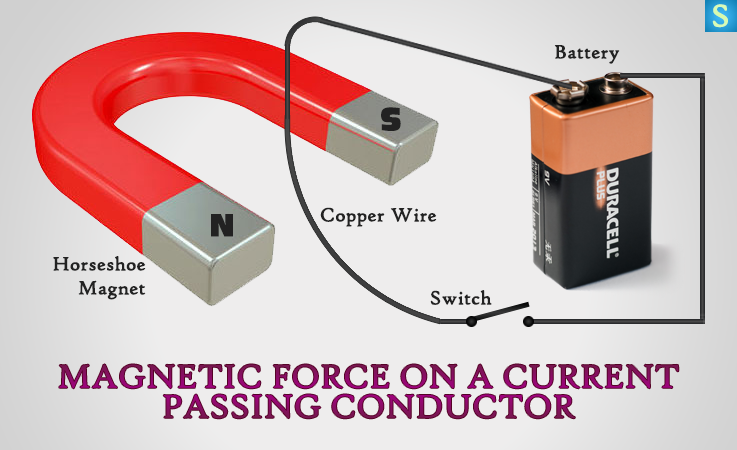
Copper wire moves outward from the horseshoe magnet in the first stage. Then the both 2nd & 3rd stages reveal that the copper wire moves into the horseshoe magnet. The wire moves because the permanent magnet’s magnetic field reacts to the electromagnetic field in the wire.
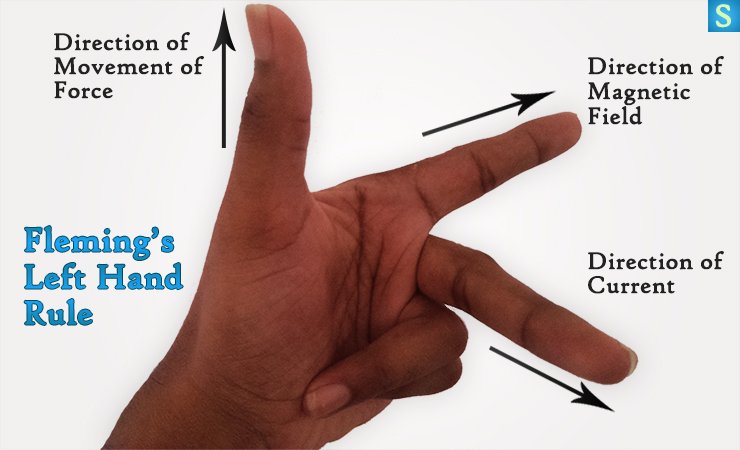
You could remember the direction of this movement with the Fleming’s left hand rule.
Fleming’s Left Hand Rule Is …
Fleming’s left hand rule simply helps you find the direction of the reaction.
Hold the first, second and thumb fingers of your left hand so that they are at right angle to each other.
- First finger should points in the direction of the magnetic field. (From N to S)
- Rotate your hand around the first finger till second finger points in the direction of electric current.
- Now your thumb automagically points in the direction of wire movement.
You’ll realize the Fleming’s Left Hand Rule is correct, if you were to do the above steps in the previous experiment.
Direct Current Motor
A simple DC motor contains a rotatable coil in between the poles of a permanent curved magnet. The current from the battery flows through brushes to the commutator which is connected to the ends of the coil. The commutator is a copper ring cut into two halves.
By using Fleming’s left hand rule for the arm AB of the coil, you’ll find that the wire moves upward so that the coil turns clockwise.
When the arm CD of the coil comes near the North-pole of the magnet, the half ring (Commutator) connected to CD is in contact with the brush connected to the negative terminal of the battery. Therefore current is coming out towards you. This means that the force on CD is upwards and the coil (electromagnet) will turn clockwise continuously.
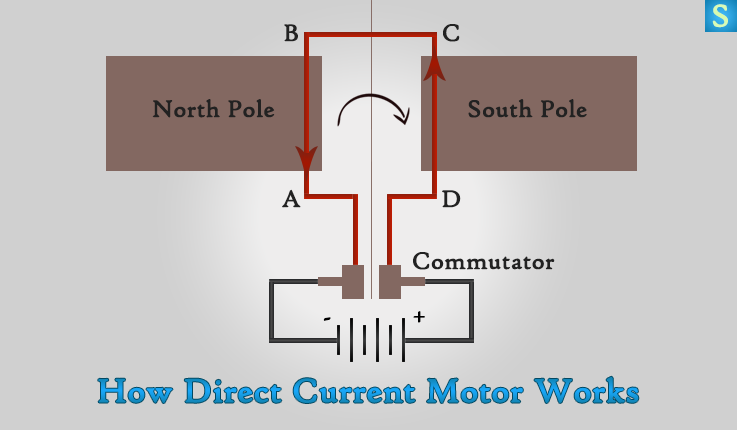
To make sure the coil turning round in the same direction you have to change the direction of the current in the coil. In other words, you have to reverse the current in the coil. Because of this, the current through the coil in a DC motor is reversed in every half turn by a commutator.
What would you think will happen if you were to reverse the terminals of the battery?
It would change the direction of the rotor rotates!
Chemical Effects of Electricity
Chemical reactions happens when electricity passes through various conducting liquids. This is known as chemical effects of electricity.
Yes, I specifically said “conducting liquids” because not all liquids conduct electricity. Let me show you if it’s the truth using below experiment.
What you need,
- Beaker with 2 electrodes
- Battery & Switch
- Light Bulb
- Crocodile Clips
- Distilled Water, NaCl Solution, Vinegar, Paraffin and HCL
Connect the circuit to two electrodes in the beaker. Pour one of the liquids into the beaker. Switch ON and note whether the bulb is glowing. Now remove the liquid and wash the apparatus properly.
Try the same with distilled water, NaCl solution, Vinegar, Paraffin and HCl.
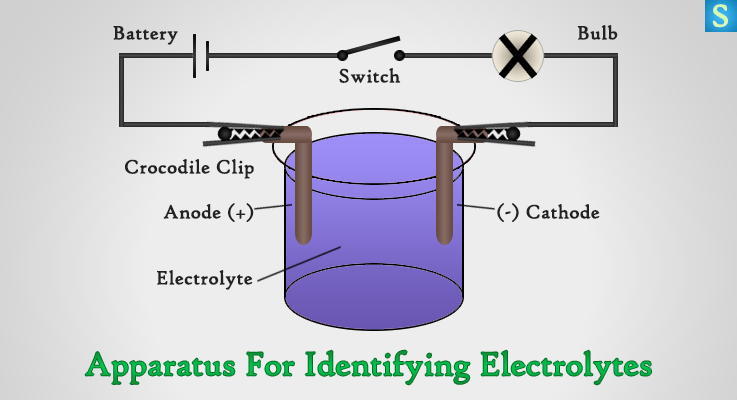
You will experience that the bulb glows when the solution inside the beaker is NaCl solution, Vinegar or HCl. Bulb does not glow when the liquid either of these, distilled water and Paraffin. It is clear that the bulb will glow when liquids conduct electricity, and the bulb will not glow when they do not conduct electricity.
Few Points on Chemical Effects of Electric Current
- Substances which are producing solutions that conduct electricity are called electrolytes. Conductors dipped in electrolytes are called electrodes.
- Anode (+) is the electrode at which electric current enters to the electrolyte, while the electrode at which it leaves the electrolyte is called the cathode (-).
- Chemical reactions occur when electricity is passed through a liquid. These chemical reactions take place at the cathode and anode. Therefore chemical changes occur in the liquid too. This is known as electrolysis.
- Voltameter is the name given for the apparatus used for electrolysis, but it’s not a voltmeter. The voltameter is named according to the electrolyte and electrodes used. For example, if the electrodes are copper and the electrolyte is a solution of copper salt, the voltameter is named as a Copper Voltameter.
Electrolysis of Water: What Happens?
What you need,
- Beaker filled with water
- 2 Platinum Electrodes
- 2 Glass Tubes
- Small amount of Sulphuric Acid
- Battery, Switch & Wire
Pass electric current through water using a Water Voltameter with two platinum electrodes connected to a battery. Since water is a poor conductor of electricity, add a small amount of Sulphuric acid to make it an electrolyte.
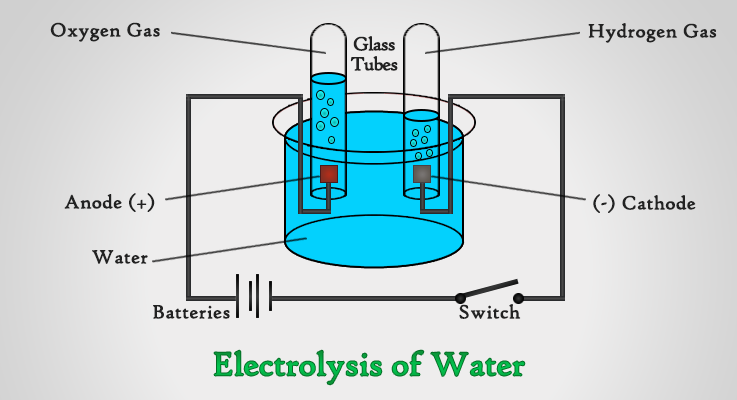
Air bubbles appear at both (+) Anode and (-) Cathode tubes. If you were to collect and test these gases, you will find that they are Oxygen at the anode and Hydrogen at the cathode.
In other words, water has decomposed into Oxygen and Hydrogen, by the chemical effect of electric current. The volume of the gases produced are in the ratio of 1:2, because water is made up of 1 Oxygen atom and 2 Hydrogen atoms.
Electrolysis of Copper Sulphate: What Happens?
What you need,
- Copper Sulphate solution filled beaker
- 2 Copper Electrodes
- 2 Crocodile Clips
- Battery, Switch & Wire
Setup the Copper Voltameter as shown in image and connect it to the circuit. Label the 2 electrodes and find the mass of each (upto 0.1 gram), before putting them into the electrolyte. Now pass a 0.5 Ampere electric current for about 30 minutes. Remove the electrodes, dry them carefully and find the new mass of both electrodes.
You will realize that the anode has lost in mass while the cathode has gained in mass by an amount equal to the mass lost by the cathode. It is clear when an electric current flows through the Copper Voltameter a chemical reaction occurs at the electrodes.
Cathode has gained in mass while also getting covered with a shiny copper coat. Therefore, you can say the cathode is electroplated with copper.
Do you know, the mass of metal deposited on the cathode in the electrolysis process is proportional to the amount of electric current passed through the voltameter and the time of passing the current. The scientist Michael Faraday discovered this for the first time.
How exactly conduction of electricity occurs through an electrolyte?
Well, the chemical formula of Copper Sulphate is CuSO4. Copper Sulphate splits into two charged particles Cu++ and SO42- when dissolved in water. These are known as Ions. The ions formed this way are the charge carriers for the passage of electricity through the electrolyte.
A Copper atom (Cu) becomes a positive Cu++ ion by losing two electrons. Due to the fact that unlike charges attract each other, this Cu++ ion drifts towards the negative cathode and gains two electrons to become a Copper atom again. This gets deposited on the cathode. The SO4– ion drifts towards the positive anode and completes the circuit.
Electroplating
Plating of one metal with another metal by electrolysis is known as electroplating. Let’s do another experiment using the same Copper Voltameter designed for previous experiment.
Replace the Copper cathode with a clean metal object such as a Door Key, Spoon etc. and pass a 0.5 Ampere of electric current for 30 minutes. Take out the object and have a look at it. You will recognize that the metal object is electroplated with Copper.
In a similar way to Copper plating in the above experiment, if the spoons were used as the Cathode of voltameter containing silver electrolyte, they can be silver plated. Chromium plating for bicycles and cars is also achieved using a similar method, but with a Chromium Salt electrolyte.
Uses of Electrolysis
As with other 2 effects of electric current, this effect too has a bunch of uses. Here are 3 benefits of chemical effects of electric current.
1. Electroplating
Most common Kitchen appliances are Nikel plated. Vehicle parts are Chromium plated to protect them from rusting.
2. Refining of Copper
Copper transferred to the Cathode is totally pure in Copper plating, even if the Anode may have been made of impure Copper. This process is used to produce Pure Copper for electric cables.
3. Extraction of Sodium and Aluminium
These metals are obtained by electrolysis of common salt for Sodium and Aluminium Oxide for Aluminium.
You know, electrolysis is used in the beauty industry as well as for hair removal which is another benefit.
I know it’s been a really long post, but now you know the main 3 effects of electric current, their uses and how they work at the end of it with so much details. Did something important missed in this article? Comment below and also consider sharing with your friends and teachers.





Dear SHYAM CHATHURANGA,
Thank you for this excellent article. With the images and sketches, it makes understanding what I had read in college days more meaningful. Your hard work and efforts in putting this is very much appreciated.
Regards,
Mahesh Khera
Thanks for the happy and warm words Mahesh. It’s feeling great to be able to help you. 💓🙏
Excellent article. Very helpful 🤗
I am in 7th class and understood my levelled part of this article completely
thank you!